The UK’s medicines regulator has issued a major recall of a commonly prescribed antidepressant after a serious packaging error was discovered by a patient.
The Medicines and Healthcare products Regulatory Agency (MHRA) announced that more than 80,000 packs of medication are being withdrawn as a precaution following the incident.
What Went Wrong?
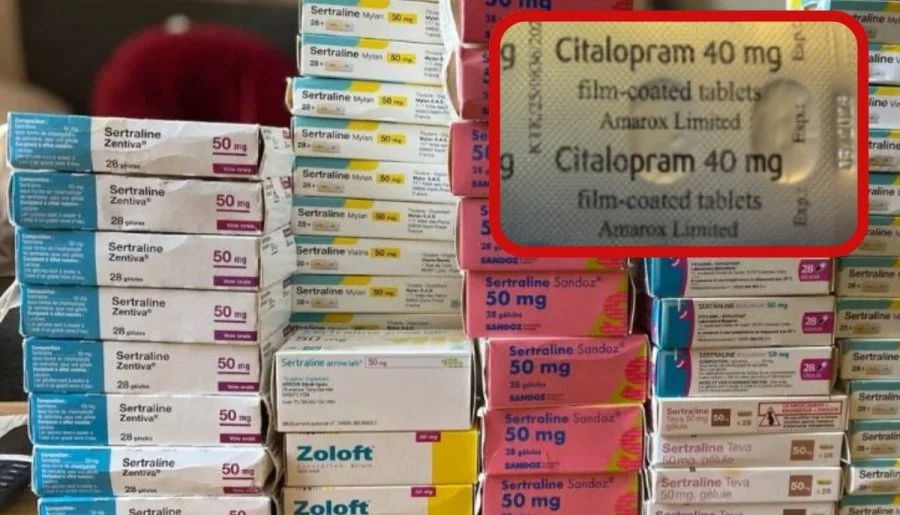
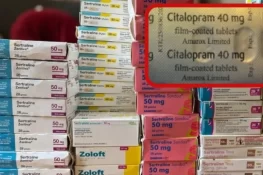
The recall was triggered when a patient, who had been prescribed Sertraline 100mg tablets, opened a sealed package and instead found Citalopram 40mg tablets inside.
Both drugs are widely used antidepressants, but they are prescribed in different doses and for specific patient needs. Receiving the wrong medication—even within the same drug class—can lead to unintended side effects or reduced treatment effectiveness.
According to the MHRA, the issue likely occurred during the secondary packaging stage, where blister packs are placed into cartons. Notably, both medications were manufactured at the same production facility.
Recall and Safety Measures
Following the report, UK-based pharmaceutical company Amarox initiated a “precautionary recall” of the affected batch of sertraline.
The MHRA has advised:
- Pharmacies to immediately stop supplying the affected batch
- Pharmacists to contact patients who may have received the product
- Patients to check their medication packaging carefully
Anyone who believes they may have taken the wrong tablets or is experiencing unusual side effects is urged to seek medical advice immediately.
Understanding the Medications Involved
Both sertraline and citalopram belong to a class of drugs known as Selective Serotonin Reuptake Inhibitors (SSRIs). These medications are commonly prescribed to treat:
- Depression
- Anxiety disorders
- Panic disorders
- Other mental health conditions
They work by increasing levels of serotonin in the brain, a chemical associated with mood regulation.
However, despite their similarities, the dosage and patient suitability differ, making accurate dispensing critical.
Why This Matters
Medication errors, even rare ones, can have serious consequences. This incident highlights the importance of quality control in pharmaceutical packaging and the need for vigilance by both healthcare providers and patients.
While the recall is described as precautionary, it underscores how even small errors in the supply chain can affect thousands of patients.
The MHRA’s swift action aims to prevent any potential harm from this packaging mix-up. Patients are encouraged to remain alert, verify their medications, and consult healthcare professionals if they notice anything unusual.